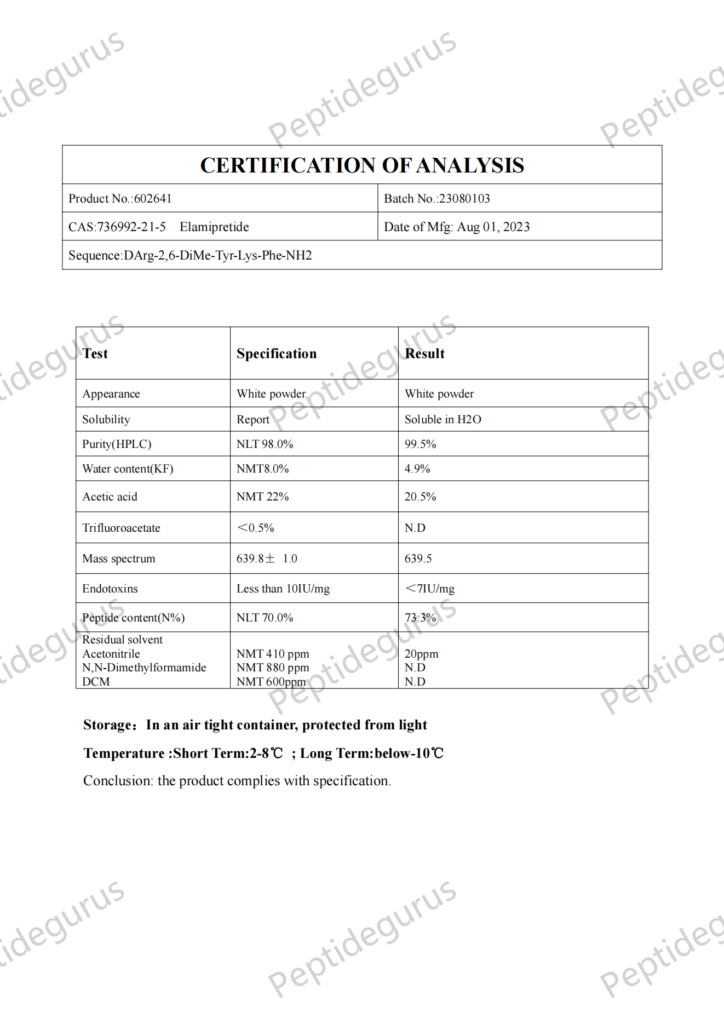
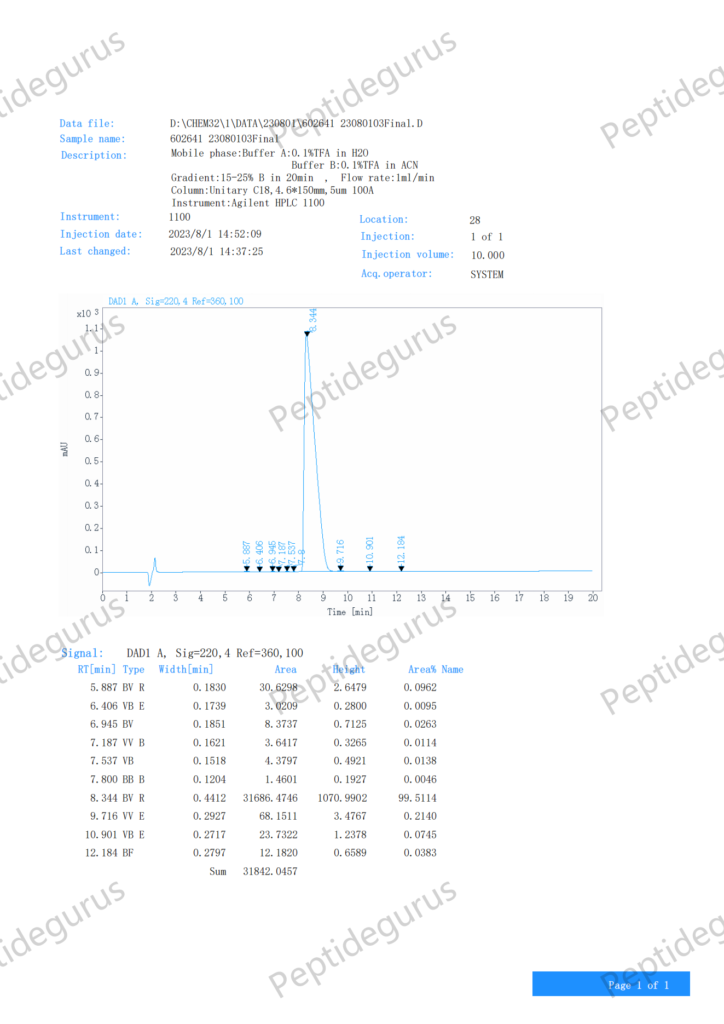
Wat is SS-31?
SS-31 (elamipretide) is a small, aromatic peptide that easily penetrates cell and organelle membranes.It is thought to interfere with the production of reactive oxygen species (ROS or free radicals) and promote energy production in cells by stabilizing the enzyme cardiolipin within mitochondria. Cardiolipin is part of the inner mitochondrial membrane where it acts as a fundamental component of the electron transport chain, the machinery by which most energy need for cellular functioning is produced.
Dysfunction of cardiolipin has been implicated as contributing to the pathology of a number of diseases including Alzheimer’s disease, Parkinson’s disease, nonalcoholic fatty liver disease, diabetes, heart failure, HIV, cancer, chronic fatigue syndrome, and more. Cardiolipin is thought to be a major component of mitochondrial myopathy, which isn’t a single disease but rather is a group of neuromuscular disorders caused by damage to mitochondria. Mitochondrial myopathy is characterized by everything from muscle weakness and exercise intolerance to heart failure, seizures, and dementia. SS-31 is the first peptide to ever undergo clinical trials as a potential treatment for mitochondrial myopathy.
Mitochondria -verbetering
Primary mitochondrial diseases (PMDs) are among the most common inherited conditions in the world. They are caused by dysfunction in the energy-producing apparatus of mitochondria. Symptoms vary greatly between forms of the disease, but the most susceptible organ systems are those with high energy demands (e.g. nervous system, heart, kidney, etc.). Muscle involvement and exercise intolerance are almost universal in mitochondrial disorders. Common symptoms include easy fatigue, exercise intolerance, and seizures.
PMDs, and mitochondrial diseases in general, are primarily characterized by disturbances in the production of ATP. ATP acts as the energy currency of the cell and is necessary to almost every cell function. Stabilizing ATP production in the setting of mitochondrial disease has long been a goal of the medical profession. With the development of SS-31, that goal may final have been realized.
The first evidence that SS-31 could restore energy production in PMDs came from animal studies. In that research, rats who had suffered ischemia-perfusion injury (a non-genetic cause of mitochondrial disease) of the kidney were given SS-31. The peptide protected kidney structure, accelerated recovery of ATP production, and reduced cell death and necrosis within the kidney[1]. Subsequent studies in mice showed that SS-31 interacted with cardiolipin in the inner mitochondria membrane and revealed that the peptide could reduce symptoms of mitochondrial disease regardless of etiology. There is also evidence that it can improve mitochondrial dysfunction that results from age[2]–[4]. From these findings, it was relatively simple to convince the FDA to grant orphan drug status to SS-31 and pave the way to clinical trials.
In phase II trials in humans, SS-31 increased exercise performance after just 5 days of treatment and showed no safety concerns or prominent side effects[5]. Unfortunately, phase III trials failed to produce convincing evidence of SS-31’s clinical utility[6]. That said, there is good reason to believe that the trial endpoints simply weren’t appropriate and that additional work will result in the peptide being approved for the treatment of certain mitochondrial conditions. According to Dr. Bruce Cohen, director of the Neurodevelopment Science Center at Akron Children’s Hospital, the results for prior phase II clinical trials were very encouraging and so it is not time to give up. Rather, he notes, SS-31 should spur interest in this particular area and bring other big pharma research to the table[7]. It appears that that is already happening as the company that first brought SS-31 to clinical trials is planning to move forward with trials of a derivative of SS-31 as well as trials investigating other endpoints for SS-31 treatment[6].
As of right now, SS-31 is being tested in a number of different human diseases and under a number of different trial models. The peptide is considered safe to use in humans, so it can also be prescribed by doctors under compassionate care exceptions to patients who have no other treatment options. The peptide will likely become part of mainstream medical care for a number of conditions in the near future, but even now it is available to people who need it while clinical trial work is ongoing.Ischemie
Perhaps the most compelling secondary application of SS-31 is in the treatment of heart failure. It has long been known that heart failure causes negative changes to the function of mitochondria and that these changes, in a kind of destructive cycle, cause heart failure to worsen. Research in human heart tissue treated with SS-31 shows significant improvements in mitochondrial oxygen flux and activity of specific components involved in the production of ATP. This particular study was carried out in a manner that precluded cardiolipin restructuring, however, suggesting that SS-31 may have a second mechanism of action on mitochondrial function that needs to be explored[4]. This finding has actually been replicated in a number of research studies, strengthening the idea that SS-31 is not just useful for restoring ATP production via cardiolipin interaction. The peptide is actively being investigated for its ability to alter the production of reactive oxygen species and improve mitochondrial function in both acute and chronic use situations.
Studies in dogs, for instance, show that chronic treatment with SS-31 can improve left ventricular function in the setting of advanced heart failure. Measures of mitochondrial respiration and maximum ATP synthesis correlated well in this study with overall improvement in left ventricular function implying that SS-31 could be an effective long-term treatment for improving energy dynamics and reducing cardiac remodeling in advanced heart failure[8].
Trials exploring the use of SS-31 in ST-segment elevation myocardial infarction (heart attack) found that the peptide can drastically reduce levels of HtrA2. HtrA2 is a measure of cardiomyocyte apoptosis. These results suggest that SS-31 may be useful in the context of acute heart attack to reduce the extent of injury and preserve cardiac tissue[9].
One role of mitochondrial-targeted therapy in heart failure: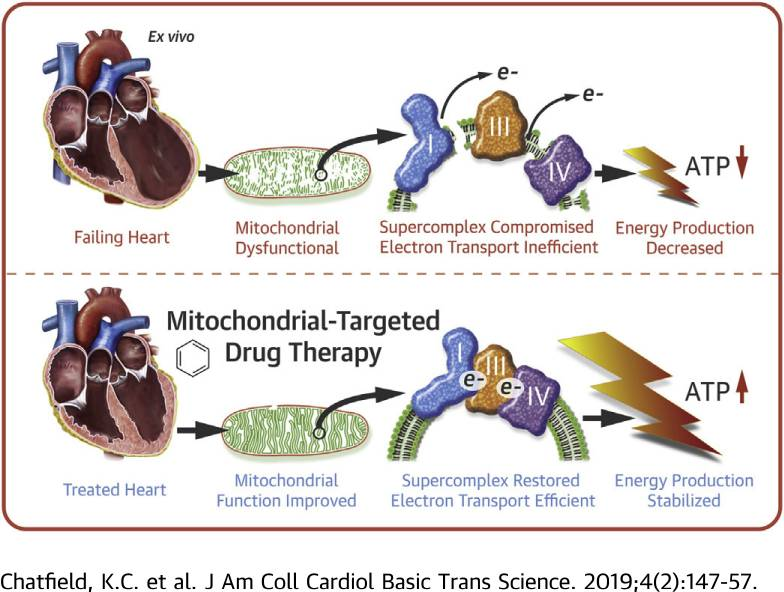 Bron:Pubch
Bron:Pubch
Diabetes
Diabetes, hoewel schijnbaar veroorzaakt door een eenvoudige ontoereikendheid in insulinesecretie of functie, is een complexe toestand met meerdere pathofysiologische manifestaties. In de afgelopen jaren is er een groeiende interesse geweest in de rol van mitochondriale stoornissen in de pathogenese van de ziekte, met name bij diabetes type 2. Het behandelen van mitochondriale disfunctie zou dus een manier zijn om enkele van de langetermijngevolgen van diabetes zoals oxidatieve schade aan kleine vaten te verbeteren. In een studie bij mensen die SS-31 kregen, werd een duidelijke afname van de productie van reactieve zuurstofspecies waargenomen. Dit suggereert dat SS-31 kan helpen om de oxidatieve schade te verminderen die meestal gepaard gaat met mitochondriale disfunctie en daarom de progressie van microvasculaire aandoeningen bij diabetes type 2 kan vertragen of stoppen. Deze hypothese wordt verder bevestigd door de bevinding, in dezelfde studie, dat SS-31 niveaus van SIRT1 verhoogde. SIRT1 -niveaus zijn geassocieerd met verbeterde insulinegevoeligheid en verminderde ontsteking bij diabetes type 2 [10].Vermindert ontsteking
A theme throughout the sections above is inflammation and the ability of SS-31 to reduce it. In particular, SS-31 appears to be a potent regulator of reactive oxygen species (free radicals) and thus helps to reduce the serious oxidative stress that arises from long-term illness such as diabetes, heart disease, and more. Research in cell cultures suggests that SS-31 reduces inflammation and oxidative stress by reducing expression of FIS1[11]. FIS1 is a mitochondrial protein that is important for mitochondrial growth and division. Elevated levels of FIS1 have been observed in a number of neurodegenerative diseases as well as a variety of cancers and are thought to be evidence of dysfunctional mitochondrial division secondary to dysfunction and inflammation.
There is also good evidence from mouse models to show that SS-31 reduces levels of the inflammatory cytokine CD-36, reduces expression of activated MnSOD, suppresses NADPH oxidase function, and inhibits NF-kappaB p65[12]. All of these are markers of high oxidative stress, so reducing their levels is indicative of reduced free radical production and an improved inflammatory status in the cell. NF-kappaB expression, in particular, is heavily associated with cellular inflammation and is chronically active in a number of inflammatory diseases like rheumatoid arthritis and inflammatory bowel disease. With SS-31, mitochondria do not undergo inflammasome activation, which is to say they don’t convert from the primary production of ATP to primarily producing ROS.
Inflammasome activation is avoided and normal mitochondrial function is preserved in the setting of SS-31 administration: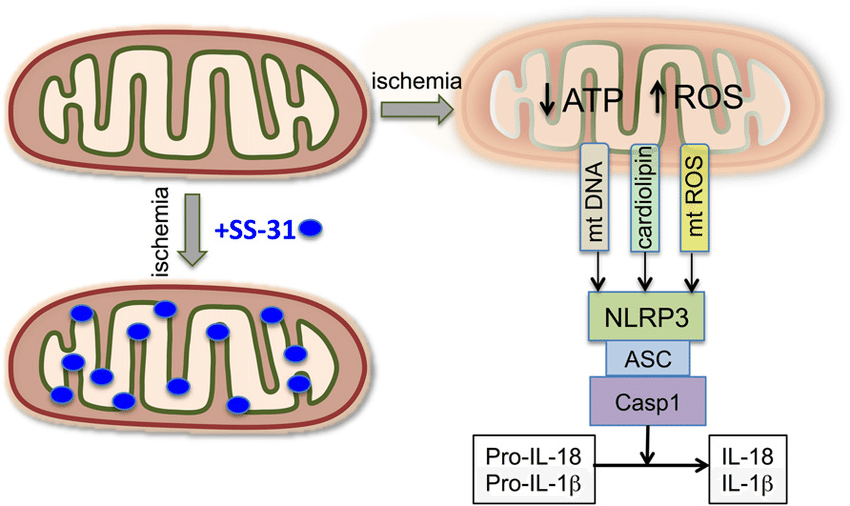 Bron:Onderzoek
Bron:Onderzoek
SS-31 Samenvatting
Hoewel SS-31 oorspronkelijk van belang was omdat wordt gedacht dat het de mitochondriale functie reguleert bij de setting van mitochondriale ziekte, is er ook goed bewijs dat het peptide door mitochondria geïnduceerde ontsteking kan reguleren. Er is veel actieve interesse in het gebruik van SS-31 om de mitochondriale functie en dus de algehele productie van energie via ATP-synthese te verbeteren. Hoewel de initiële fase III -proeven niet succesvol waren, wordt gedacht dat dit meer het gevolg kan zijn van de eindpunten gemeten in tegenstelling tot een echt falen van het peptide om enig effect te hebben. Momenteel zijn er lopende fase II-onderzoeken en geplande fase III-onderzoeken om SS-31 te testen in verschillende ziektetoestanden en met een verscheidenheid aan verschillende uitkomstmaten. SS-31 kan heel goed de sleutel bieden om mitochondriale disfunctie bij verschillende ziekten te begrijpen en kan dus nuttig zijn bij het ontwerpen van geavanceerde behandelingen voor de ziekte van Alzheimer, de ziekte van Parkinson, hartaandoeningen, diabetes, nierziekte en meer.Artikelauteur
De bovenstaande literatuur werd onderzocht, bewerkt en georganiseerd door Dr. E. Logan, M.D. Dr. E. Logan behaalde een doctoraat vanCase Western Reserve University School of Medicineen een B.S. in moleculaire biologie.Wetenschappelijke tijdschrift Auteur
In zijn werk om behandelingen te ontdekken voor patiënten met mitochondriale ziekten, is Dr. Cohen een van 's werelds toonaangevende experts geworden. Hoewel er geen behandelingen zijn voor mitochondriale ziekten, zoekt Dr. Cohen er elke dag naar een. Als gevolg van zijn werk en de toewijding van ons ziekenhuis om hem te helpen een infrastructuur op te bouwen voor het uitvoeren van mitochondriale klinische proeven, is Akron Children's een van de toonaangevende instellingen van het land die behandelingen onderzoekt. Voordat hij bij Akron Children's was, diende Dr. Cohen als chief van pediatrische neurologie voor Cleveland Clinic. Hij behaalde zijn medische graad aan het Albert Einstein College of Medicine en voltooide zijn pediatrische residentie in het kinderziekenhuis van Philadelphia, pediatrische neurologie residentie in het Columbia Presbyterian Medical Center en zijn American Cancer Society Neuro-oncology Fellowship in Philadelphia.Bruce H. Cohen, M.D.wordt waarnaar wordt verwezen als een van de toonaangevende wetenschappers die betrokken zijn bij het onderzoek en de ontwikkeling van SS-31. Op geen enkele manier is deze arts/wetenschapper de aankoop, verkoop of het gebruik van dit product om welke reden dan ook goed te goedkeuren of te bepleiten. Er is geen aansluiting of relatie, geïmpliceerd of anderszins, tussen
Peptidegoeroesen deze dokter. Het doel van het citeren van de arts is om de uitputtende onderzoeks- en ontwikkelingsinspanningen van de wetenschappers die dit peptide bestuderen te erkennen, te erkennen en te crediteren. Dr.Cohen staat vermeld in [5] onder de citaten van de verwezen.