Free (1) 30 ml Bacteriostatic Water
with qualified orders over $500 USD.
(excludes capsule products, cosmetic peptides, promo codes and shipping)
Product Usage: This PRODUCT IS INTENDED AS A RESEARCH CHEMICAL ONLY. This designation allows the use of research chemicals strictly for in vitro testing and laboratory experimentation only. All product information available on this website is for educational purposes only. Bodily introduction of any kind into humans or animals is strictly forbidden by law. This product should only be handled by licensed, qualified professionals. This product is not a drug, food, or cosmetic and may not be misbranded, misused or mislabled as a drug, food or cosmetic.
Ovagen
Ovagen is a tripeptide bioregulator that should not be confused with the similarly named Ovagen that contains ovine (sheep) follicle stimulating hormone (FSH). The two products are not the same and have entirely different functions. While the FSH derivative promotes ovulation, particularly in women suffering from conditions like polycystic ovarian syndrome (PCOS), the tripeptide is a liver function regulator. Throughout this article, Ovagen will refer to the tripeptide bioregulator.
Ovagen is a Khavinson peptide, developed along with a number of other peptides, by Dr. Vladimir Khavinson. Like other bioregulators, Ovagen is able to cross cell and nuclear membranes to directly regulate DNA structure and transcription patterns. Also, like most other bioregulatory peptides, Ovagen has tissue-specific effects. The Glu-Asp-Leu peptide primarily serves to regulate and normalize liver and digestive tract function, though there is some evidence to suggest it may also be useful in controlling the reproduction of the HIV virus. This latter finding has made Ovagen of interest to HIV researchers looking to understand the virus lifecycle.
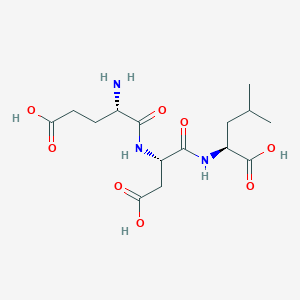
Ovagen Structure
Amino Acid Sequence: Glu-Asp-Leu (EDL)
Molecular Formula: C15H25N3O8
Molecular Weight: 375.37 g/mol
PubChem CID: 444128
Synonyms: EDL, glutamyl-aspartyl-leucine, SCHEMBL5329396, 1a30, CHEBI:137252
Ovagen and the Liver
According to research from Dr. Khavinson, Ovagen is a bioregulator of the GI tract and liver. In the liver, it has been found to promote cell proliferation and prevent scarring and fibrotic changes that can lead to cirrhosis. In the GI tract, Ovagen helps to boost mucosal barrier function and reduces complications from antibiotic treatment, environmental insults, chemotherapy, malnutrition, and more.
Like most bioregulators, the most robust effects of Ovagen are seen in older individuals. The peptide appears to roll back DNA changes that occur with age, helping to reset the DNA in liver fibroblasts and GI mucosal cells to a more youthful state. In this more youthful state, the DNA is less condensed and therefore more genes are available for transcription. This results in a more functional cell that is less senescent and therefore “healthier.”
Research shows that Ovagen is well tolerate and may be useful in normalizing the function of the liver and GI tract in a number of different inflammatory and disease conditions. It may also be useful in the postoperative setting, during long-term antibiotic therapy, for overcoming the side effects of cancer treatments, and even as a diabetes preventative. Research on the GI and liver effects of Ovagen is ongoing.
Ovagen and HIV
The HIV-1 protease is an enzyme necessary for the HIV virus to survive. It functions to cleave newly synthesized proteins that make up the mature HIV virion. This infectious form of the HIV virus will not function without the HIV-1 protease. In other words, the enzyme is essential for the HIV virus to be able to infect more cells. Without the protease, the HIV virus produces non-infectious progeny and dies out. A number of HIV protease inhibitors are currently FDA approved and used in treatment. Unfortunately, the virus’s high mutation rate results in relatively rapid formation of resistance.
Research on Ovagen reveals that it is an effective HIV-1 protease inhibitor. In fact, it is one of the smallest and most potent protease inhibitors known with an effective concentration of just 50 microM. Unlike other protease inhibitors, Ovagen is highly soluble in water[1]. This latter fact makes it easier to administer.
Ovagen Summary
Ovagen is a tripeptide bioregulator with primary effects in the liver and GI tract. Though research on this Khavinson peptide is limited at this point, it has shown promise as a potential anti-aging peptide in the liver and GI tract. Ovagen reduces long-term fibrosis in the liver and helps to protect the GI mucosal layer from the effects of antibiotics, environmental toxins, and even chemotherapy. There is also interest in the ability of Ovagen to inhibit the replication of HIV.
Article Author
The above literature was researched, edited and organized by Dr. E. Logan, M.D. Dr. E. Logan holds a doctorate degree from Case Western Reserve University School of Medicine and a B.S. in molecular biology.
Scientific Journal Author

Prof. Vladimir Khavinson is being referenced as one of the leading scientists involved in the research and development of Ovagen. In no way is this doctor/scientist endorsing or advocating the purchase, sale, or use of this product for any reason. There is no affiliation or relationship, implied or otherwise, between
Referenced Citations
ALL ARTICLES AND PRODUCT INFORMATION PROVIDED ON THIS WEBSITE ARE FOR INFORMATIONAL AND EDUCATIONAL PURPOSES ONLY.
The products offered on this website are furnished for in-vitro studies only. In-vitro studies (Latin: in glass) are performed outside of the body. These products are not medicines or drugs and have not been approved by the FDA to prevent, treat or cure any medical condition, ailment or disease. Bodily introduction of any kind into humans or animals is strictly forbidden by law.