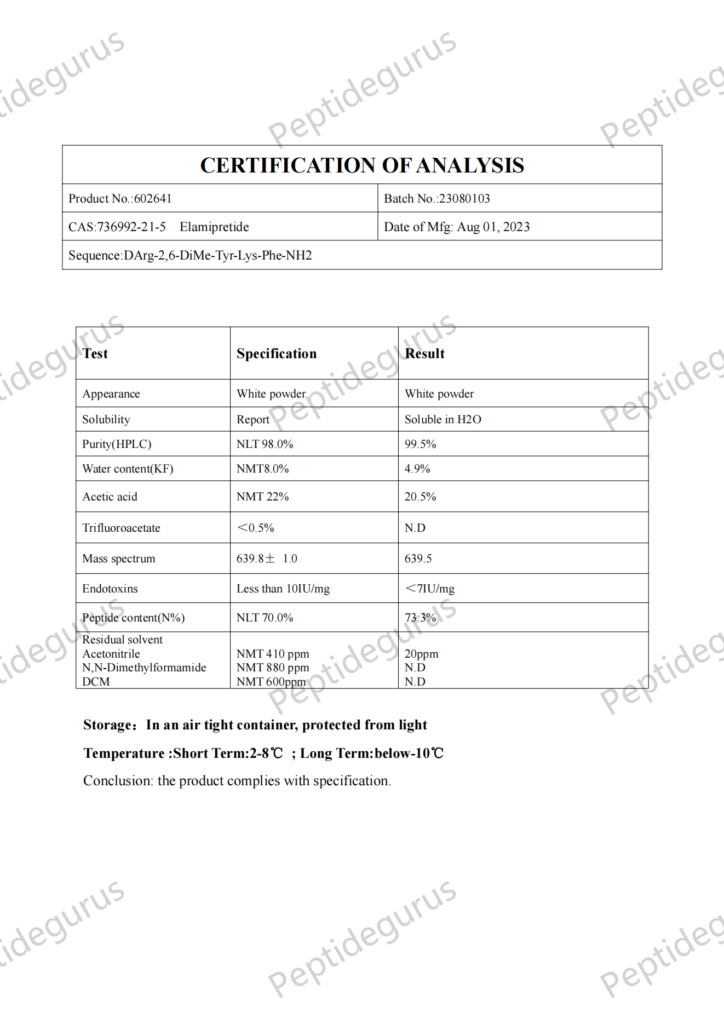
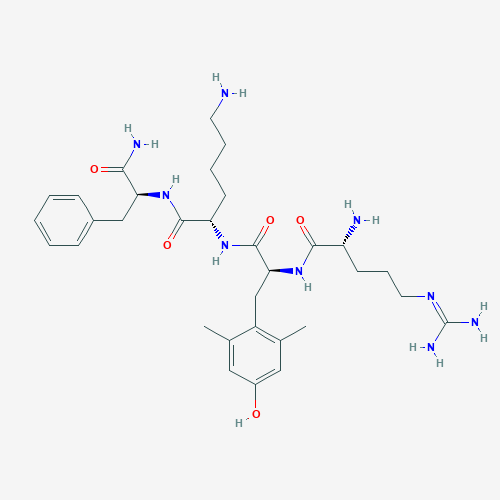
What is SS-31?
SS-31 (elamipretide) is a small, aromatic peptide that easily penetrates cell and organelle membranes.It is thought to interfere with the production of reactive oxygen species (ROS or free radicals) and promote energy production in cells by stabilizing the enzyme cardiolipin within mitochondria. Cardiolipin is part of the inner mitochondrial membrane where it acts as a fundamental component of the electron transport chain, the machinery by which most energy need for cellular functioning is produced.
Dysfunction of cardiolipin has been implicated as contributing to the pathology of a number of diseases including Alzheimer’s disease, Parkinson’s disease, nonalcoholic fatty liver disease, diabetes, heart failure, HIV, cancer, chronic fatigue syndrome, and more. Cardiolipin is thought to be a major component of mitochondrial myopathy, which isn’t a single disease but rather is a group of neuromuscular disorders caused by damage to mitochondria. Mitochondrial myopathy is characterized by everything from muscle weakness and exercise intolerance to heart failure, seizures, and dementia. SS-31 is the first peptide to ever undergo clinical trials as a potential treatment for mitochondrial myopathy.
Mitochondria Improvement
Primary mitochondrial diseases (PMDs) are among the most common inherited conditions in the world. They are caused by dysfunction in the energy-producing apparatus of mitochondria. Symptoms vary greatly between forms of the disease, but the most susceptible organ systems are those with high energy demands (e.g. nervous system, heart, kidney, etc.). Muscle involvement and exercise intolerance are almost universal in mitochondrial disorders. Common symptoms include easy fatigue, exercise intolerance, and seizures.
PMDs, and mitochondrial diseases in general, are primarily characterized by disturbances in the production of ATP. ATP acts as the energy currency of the cell and is necessary to almost every cell function. Stabilizing ATP production in the setting of mitochondrial disease has long been a goal of the medical profession. With the development of SS-31, that goal may final have been realized.
The first evidence that SS-31 could restore energy production in PMDs came from animal studies. In that research, rats who had suffered ischemia-perfusion injury (a non-genetic cause of mitochondrial disease) of the kidney were given SS-31. The peptide protected kidney structure, accelerated recovery of ATP production, and reduced cell death and necrosis within the kidney[1]. Subsequent studies in mice showed that SS-31 interacted with cardiolipin in the inner mitochondria membrane and revealed that the peptide could reduce symptoms of mitochondrial disease regardless of etiology. There is also evidence that it can improve mitochondrial dysfunction that results from age[2]–[4]. From these findings, it was relatively simple to convince the FDA to grant orphan drug status to SS-31 and pave the way to clinical trials.
In phase II trials in humans, SS-31 increased exercise performance after just 5 days of treatment and showed no safety concerns or prominent side effects[5]. Unfortunately, phase III trials failed to produce convincing evidence of SS-31’s clinical utility[6]. That said, there is good reason to believe that the trial endpoints simply weren’t appropriate and that additional work will result in the peptide being approved for the treatment of certain mitochondrial conditions. According to Dr. Bruce Cohen, director of the Neurodevelopment Science Center at Akron Children’s Hospital, the results for prior phase II clinical trials were very encouraging and so it is not time to give up. Rather, he notes, SS-31 should spur interest in this particular area and bring other big pharma research to the table[7]. It appears that that is already happening as the company that first brought SS-31 to clinical trials is planning to move forward with trials of a derivative of SS-31 as well as trials investigating other endpoints for SS-31 treatment[6].
As of right now, SS-31 is being tested in a number of different human diseases and under a number of different trial models. The peptide is considered safe to use in humans, so it can also be prescribed by doctors under compassionate care exceptions to patients who have no other treatment options. The peptide will likely become part of mainstream medical care for a number of conditions in the near future, but even now it is available to people who need it while clinical trial work is ongoing.Ischemia
Perhaps the most compelling secondary application of SS-31 is in the treatment of heart failure. It has long been known that heart failure causes negative changes to the function of mitochondria and that these changes, in a kind of destructive cycle, cause heart failure to worsen. Research in human heart tissue treated with SS-31 shows significant improvements in mitochondrial oxygen flux and activity of specific components involved in the production of ATP. This particular study was carried out in a manner that precluded cardiolipin restructuring, however, suggesting that SS-31 may have a second mechanism of action on mitochondrial function that needs to be explored[4]. This finding has actually been replicated in a number of research studies, strengthening the idea that SS-31 is not just useful for restoring ATP production via cardiolipin interaction. The peptide is actively being investigated for its ability to alter the production of reactive oxygen species and improve mitochondrial function in both acute and chronic use situations.
Studies in dogs, for instance, show that chronic treatment with SS-31 can improve left ventricular function in the setting of advanced heart failure. Measures of mitochondrial respiration and maximum ATP synthesis correlated well in this study with overall improvement in left ventricular function implying that SS-31 could be an effective long-term treatment for improving energy dynamics and reducing cardiac remodeling in advanced heart failure[8].
Trials exploring the use of SS-31 in ST-segment elevation myocardial infarction (heart attack) found that the peptide can drastically reduce levels of HtrA2. HtrA2 is a measure of cardiomyocyte apoptosis. These results suggest that SS-31 may be useful in the context of acute heart attack to reduce the extent of injury and preserve cardiac tissue[9].
One role of mitochondrial-targeted therapy in heart failure: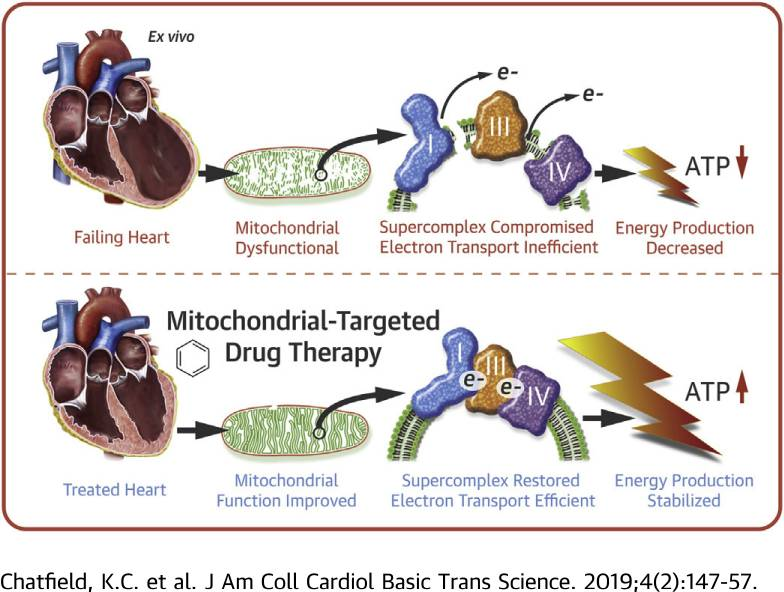 ที่มา:ผับเคม
ที่มา:ผับเคม
Diabetes
โรคเบาหวานแม้ว่าจะดูเหมือนจะเกิดจากความไม่เพียงพอในการหลั่งอินซูลินหรือการทํางาน แต่ก็เป็นภาวะที่ซับซ้อนโดยมีอาการทางพยาธิสรีรวิทยาหลายอย่าง ในช่วงไม่กี่ปีที่ผ่านมา มีความสนใจเพิ่มขึ้นในบทบาทของความบกพร่องของไมโทคอนเดรียในการเกิดโรค โดยเฉพาะอย่างยิ่งในโรคเบาหวานประเภท 2 การรักษาความผิดปกติของไมโทคอนเดรียจึงเป็นวิธีปรับปรุงผลที่ตามมาในระยะยาวของโรคเบาหวาน เช่น ความเสียหายจากปฏิกิริยาออกซิเดชันต่อหลอดเลือดขนาดเล็ก ในการศึกษาในมนุษย์ที่ได้รับ SS-31 พบว่าการผลิตออกซิเจนที่ทําปฏิกิริยาลดลงอย่างเห็นได้ชัด นี่แสดงให้เห็นว่า SS-31 สามารถช่วยลดความเสียหายจากปฏิกิริยาออกซิเดชันที่มักมาพร้อมกับความผิดปกติของไมโทคอนเดรีย และอาจชะลอหรือหยุดการลุกลามของโรคหลอดเลือดขนาดเล็กในโรคเบาหวานประเภท 2 สมมติฐานนี้ได้รับการยืนยันเพิ่มเติมจากการค้นพบในการศึกษาเดียวกันว่า SS-31 เพิ่มระดับ SIRT1 ระดับ SIRT1 สัมพันธ์กับความไวของอินซูลินที่ดีขึ้นและลดการอักเสบในโรคเบาหวานประเภท 2[10]Reduces Inflammation
A theme throughout the sections above is inflammation and the ability of SS-31 to reduce it. In particular, SS-31 appears to be a potent regulator of reactive oxygen species (free radicals) and thus helps to reduce the serious oxidative stress that arises from long-term illness such as diabetes, heart disease, and more. Research in cell cultures suggests that SS-31 reduces inflammation and oxidative stress by reducing expression of FIS1[11]. FIS1 is a mitochondrial protein that is important for mitochondrial growth and division. Elevated levels of FIS1 have been observed in a number of neurodegenerative diseases as well as a variety of cancers and are thought to be evidence of dysfunctional mitochondrial division secondary to dysfunction and inflammation.
There is also good evidence from mouse models to show that SS-31 reduces levels of the inflammatory cytokine CD-36, reduces expression of activated MnSOD, suppresses NADPH oxidase function, and inhibits NF-kappaB p65[12]. All of these are markers of high oxidative stress, so reducing their levels is indicative of reduced free radical production and an improved inflammatory status in the cell. NF-kappaB expression, in particular, is heavily associated with cellular inflammation and is chronically active in a number of inflammatory diseases like rheumatoid arthritis and inflammatory bowel disease. With SS-31, mitochondria do not undergo inflammasome activation, which is to say they don’t convert from the primary production of ATP to primarily producing ROS.
Inflammasome activation is avoided and normal mitochondrial function is preserved in the setting of SS-31 administration: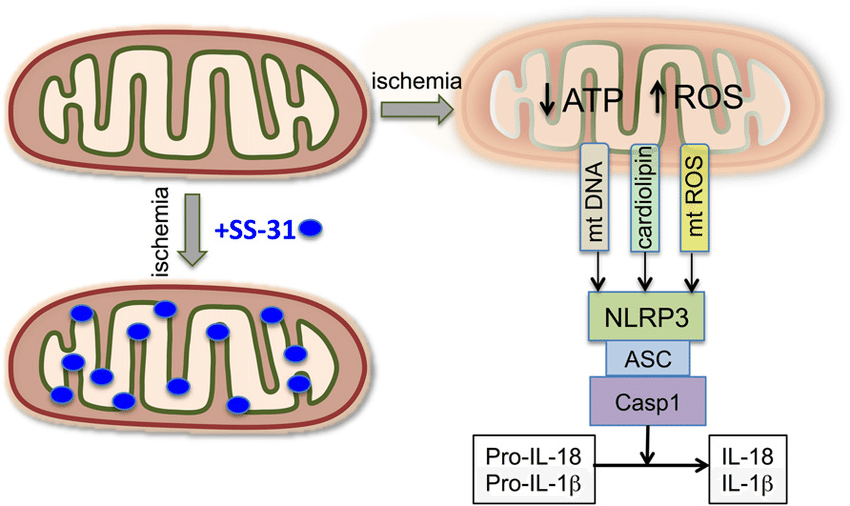 ที่มา:ResearchGate
ที่มา:ResearchGate
สรุป SS-31
แม้ว่าเดิมที SS-31 จะเป็นที่สนใจเพราะคิดว่าสามารถควบคุมการทํางานของไมโทคอนเดรียในสภาพแวดล้อมของโรคไมโทคอนเดรีย แต่ก็มีหลักฐานที่ดีว่าเปปไทด์สามารถควบคุมการอักเสบที่เกิดจากไมโทคอนเดรียได้ มีความสนใจอย่างมากในการใช้ SS-31 เพื่อปรับปรุงการทํางานของไมโทคอนเดรีย และด้วยเหตุนี้การผลิตพลังงานโดยรวมผ่านการสังเคราะห์ ATP แม้ว่าการทดลองระยะที่ 3 เบื้องต้นจะไม่ประสบความสําเร็จ แต่ก็คิดว่านี่อาจเป็นผลมาจากจุดสิ้นสุดที่วัดได้มากกว่าความล้มเหลวที่แท้จริงของเปปไทด์ที่จะมีผลใดๆ ปัจจุบันมีการทดลองระยะที่ 2 ที่กําลังดําเนินอยู่และการทดลองระยะที่ 3 ที่วางแผนไว้เพื่อทดสอบ SS-31 ในสภาวะโรคต่างๆ และด้วยการวัดผลลัพธ์ที่แตกต่างกัน SS-31 อาจเป็นกุญแจสําคัญในการทําความเข้าใจความผิดปกติของไมโทคอนเดรียในโรคต่างๆ และอาจพิสูจน์ได้ว่ามีประโยชน์ในการออกแบบการรักษาขั้นสูงสําหรับโรคอัลไซเมอร์ โรคพาร์กินสัน โรคหัวใจ เบาหวาน โรคไต และอื่นๆผู้เขียนบทความ
วรรณกรรมข้างต้นได้รับการวิจัยแก้ไขและจัดโดย Dr.. Logan, MD Dr.. Logan สําเร็จการศึกษาระดับปริญญาเอกจากคณะแพทยศาสตร์มหาวิทยาลัย Case Western Reserveและปริญญาตรีสาขาชีววิทยาโมเลกุลผู้เขียนวารสารวิทยาศาสตร์
In his work to uncover treatments for patients with mitochondrial diseases, Dr. Cohen has become one of the world’s leading experts. Although there are no cures for mitochondrial diseases, Dr. Cohen looks for one every day. As a result of his work and our hospital’s commitment to helping him build an infrastructure for conducting mitochondrial clinical trials, Akron Children’s is one of the country’s leading institutions researching treatments.Before joining Akron Children’s, Dr. Cohen served as chief of pediatric neurology for Cleveland Clinic. He received his medical degree from the Albert Einstein College of Medicine and completed his pediatric residency at Children’s Hospital of Philadelphia, pediatric neurology residency at Columbia Presbyterian Medical Center and his American Cancer Society neuro-oncology fellowship in Philadelphia.Bruce H. Cohen, M.D.is being referenced as one of the leading scientists involved in the research and development of SS-31. In no way is this doctor/scientist endorsing or advocating the purchase, sale, or use of this product for any reason. There is no affiliation or relationship, implied or otherwise, between
กูรูเปปไทด์and this doctor. The purpose of citing the doctor is to acknowledge, recognize, and credit the exhaustive research and development efforts conducted by the scientists studying this peptide. Dr.Cohen is listed in [5] under the referenced citations.