


Categorías:Péptidos de melanotan
 Secuencia: Ac-Nle-Asp(1)-His-D-Phe-Arg-Trp-LyS(1)Fórmula molecular: CsoHsN14O10Peso molecular: 1025,182 g/molPubChem ClD: 9941379Número CAS: 189691-06-3Investigación PT-141PT-141 y excitación sexual
PT-141 es un péptido único porque estimula el MC-4R, que se sabe que produce excitación sexual en el sistema nervioso central e influye en el comportamiento sexual. 2. Los estudios en ratones han demostrado que la unión del agonista al MC-4R provoca excitación sexual y aumento de la cópula. tanto en hombres como en mujeres [3], [4] Debido a que PT-141 funciona con un mecanismo diferente al de medicamentos como Viagra, es posible tratar los trastornos de la excitación sexual en tanto en hombres como en mujeres que se derivan de causas distintas a la reducción del flujo sanguíneo a los genitales.
Un estudio de hombres con disfunción eréctil (DE) que no respondieron al sildenafil (Viagra) encontró que aproximadamente un tercio experimentó una erección adecuada para tener relaciones sexuales con PT-141 (administrado mediante aerosol nasal). También hubo una fuerte respuesta dependiente de la dosis en el ensayo, lo que indica que PT-141 es realmente eficaz en ciertos casos. Esto sugiere que PT-141 podría ofrecer información sobre cómo corregir la disfunción eréctil en entornos donde el sildenafil ha fallado y puede ofrecer información sobre las causas centrales del deseo sexual hipoactivo.
Secuencia: Ac-Nle-Asp(1)-His-D-Phe-Arg-Trp-LyS(1)Fórmula molecular: CsoHsN14O10Peso molecular: 1025,182 g/molPubChem ClD: 9941379Número CAS: 189691-06-3Investigación PT-141PT-141 y excitación sexual
PT-141 es un péptido único porque estimula el MC-4R, que se sabe que produce excitación sexual en el sistema nervioso central e influye en el comportamiento sexual. 2. Los estudios en ratones han demostrado que la unión del agonista al MC-4R provoca excitación sexual y aumento de la cópula. tanto en hombres como en mujeres [3], [4] Debido a que PT-141 funciona con un mecanismo diferente al de medicamentos como Viagra, es posible tratar los trastornos de la excitación sexual en tanto en hombres como en mujeres que se derivan de causas distintas a la reducción del flujo sanguíneo a los genitales.
Un estudio de hombres con disfunción eréctil (DE) que no respondieron al sildenafil (Viagra) encontró que aproximadamente un tercio experimentó una erección adecuada para tener relaciones sexuales con PT-141 (administrado mediante aerosol nasal). También hubo una fuerte respuesta dependiente de la dosis en el ensayo, lo que indica que PT-141 es realmente eficaz en ciertos casos. Esto sugiere que PT-141 podría ofrecer información sobre cómo corregir la disfunción eréctil en entornos donde el sildenafil ha fallado y puede ofrecer información sobre las causas centrales del deseo sexual hipoactivo.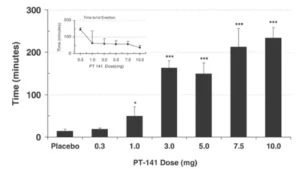 Interestingly, PT-141 was pulled from clinical trials before it reached approval for use inwomen suffering from HSDD. This is despite signs that the drug increased the number ofsatisfying sexual events per month and decreased female sexual distress scores in astatistically significant manner without any substantial side effects[5]. Many experts whotreat female sexual dysfunction (FSD) were dismayed to find the peptide was not beingadvanced despite positive results. They point to a lack of established endpoints for trialsof FSD and socio-cultural biases against women’s sexual health as the primaryroadblocks that are inhibiting approval of what they see as much-needed therapies?].They hope that greater attention will be given to the topic and that the FDA will establishmore concrete guidelines for evaluating therapies like PT-141 that can offer benefit.These experts also expressed dismay that the pharmacological treatments were nottested in conjunction with other established means of treating sexual dysfunction as theybelieve that the combination may prove synergistic and that peptides like PT-141 may beuseful for overcoming initial barriers and jump-starting psychological treatment modalities
In 2017, partly in response to the outcry against the cessation of earlier trials, Phase llReconnect trials were launched using subcutaneous injections of PT-141 for FSD. Thenewest version of PT-141, called Rekynda, may soon be available for use in the UnitedStates. lt would be legal to use PT-141 off-label, at that point, to treat both male andfemale sexual dysfunction8. These new trials have relied on the kind of modifiedendpoints that experts in FSD have touted as beneficial to seeing these kinds oftreatments approved.
PT-141 and Hemorrhage
In 2009, PT-141 was modified slightly and investigated as a potential treatment forhemorrhagic shock. Because PT-141 binds to both MC-1R and MC-4R, it reducesischemia and protects tissues against inadequate blood supply in the setting ofhypovolemic (hemorrhagic) shock. The drug, when administered intravenously, does notproduce substantial side effects. lt was last in phase llb trials. The modified version of PT-141 is referred to as PL-6983.
PT-141 and Infection
The MC-1R has been found, in a rat model of a specific fungal infection, to possessimportant anti-fungal and anti-inflammatory properties[9]. This is of particular importancebecause current anti-fungals are limited in terms of their mechanism of action and allproduce serious and treatment-limiting side effects in certain patients. Having analternative to use in the treatment of fungal infections could reduce morbidity andmortality substantially, especially in patients with immune compromise.
PT-141 and Cancer
The MC-1R receptor is an important stimulus of DNA repair pathways and thus is ofinterest in cancer treatment and prevention[1o]. Research shows that people with variantsof MC-1R are at increased risk for both basal cell and squamous cell carcinoma[10].Altered PT-141 may be able to correct the problems experienced as a result of thesevariants and prevent or treat these cancers.Direcciones de investigaciónEn este momento, PT-141 ha recibido una atención intensa y generalizada como tratamiento para la disfunción sexual. Sin embargo, existe una gran cantidad de investigaciones potenciales fuera de la disfunción sexual y la hemorragia a las que se podría aplicar el PT-141. Por ejemplo, se sabe que el MC-4R está defectuoso o falta en ciertos casos de obesidad y puede representar hasta el 6% de todos los casos de obesidad de aparición temprana. PT-141 ofrece un medio único para explorar esta causa particular de la obesidad y potencialmente ilustrar una vía de intervención. MC-1R desempeña un papel tanto en el dolor como en la inflamación, así como en la patología renal y la propagación de infecciones. Hay una gran cantidad de investigaciones disponibles sobre las que PT.141 podría ayudar a arrojar luz.
PT-141 presenta efectos secundarios mínimos, baja biodisponibilidad oral y subcutánea excelente en ratones. La dosis por kg en ratones no se ajusta a la de los humanos. El PT-141 a la venta en PeptideSciences se limita únicamente a la investigación científica y educativa, no al consumo humano. Compre PT-141 únicamente si es un investigador autorizado.Autor del artículoLa literatura anterior fue investigada, editada y organizada por el Dr. Logan, M.D. El Dr. Logar tiene un doctorado de la Facultad de Medicina de la Universidad Case Western Reserve y una licenciatura. en biología molecular.Autor de revista científicaDr. Sheryl A. Kingsberg is the chief of behavioral medicine at University Hospitals CaseMedical Center and professor in Reproductive Biology and Psychiatry at Case WesternReserve University. Her areas of clinical specialization include sexual medicine, femalesexual disorders, cognitive behavioral psychotherapy, menopause, pregnancy andpostpartum mood disorders, psychological aspects of infertility, and psychological andsexual aspects of cancer. Dr. Kingsberg’s primary research interests are in treatments forfemale sexual disorders and the psychological aspects of infertility and menopause. Sheled a randomized, placebo-controlled dose-finding trial for PT-141. She has numerouspublications in many national and international journals, sits on the editorial board ofMenopause and has authored numerous chapters on topics including perimenopauseand sexuality, oocvte donation, infertility and aging, the treatment of psychogenic erectiledysfunction and sexuality after cancer. Dr. Kingsberg received her PhD from theUniversity of South Florida in Tampa and completed her fellowshin in sexual medicine aUniversity Hospitals Case Medical Center, She is an active member in a number ofnational and international organizations including the American Psychological Associationand the American Society for Reproductive Medicine. She currently sits on the Board ofTrustees of The North American Menopause Society, and serves as the current treasurerof the Society for Assisted Reproductive Technologies. Dr. Kingsberg s a past president otThe International Society for the Study of Women’s Sexual Health.
Dr. Sheryl A. Kingsberg is being referenced as one of the leading scientists involved inthe research and development of PT-141, in no way is this doctor/scientist endorsing oradvocating the purchase, sale, or use of this product for any reason, There is no affiliationor relationship, implied or otherwise, between PEPTIDE GURUS and this doctor. Thepurpose of citing the doctor is to acknowledge, recoanize, and credit the exhaustiveresearch and development efforts conducted by the scientists studying this peptide. DrKingsbera is listed in [12l under the referenced citations.Citas referenciadas1.M.Sandrock,A.Schulz,C.Merkwitz,T.Schüneberg, K.Spanel-Borowski, and A.Ricken, “Reduction in corpora lutea number in obese melanocortin-4-receptor-deficient mice,”Reprod. Biol. Endocrinol.RBE,vol.7. p.24, Mar. 2009.
2.R.C.Rosen,L.E.Diamond, D.C.Earle,A. M. Shadiack, and P. B. MolinoffEvaluation of the safety, pharmacokinetics and pharmacodynamic effects ofsubcutaneously administered PT-141, a melanocortin receptor agonist, in healthymale subjects and in patients with an inadeguate response to Viagra,” int. J. lmpot.Res., vol.16,no.2,pp.135-142,Apr.2004.[PubMed]
3.H.Wessells,V.. Hruby, J.Hackett, G. Han, P. Balse-Srinivasan, and T. W.Vanderah,“Ac-Nle-c[Asp-His-DPhe-Arg-Trp-Lys]-NH2 induces penile erection viabrain and spinal melanocortin receptors,”Neuroscience, vol. 118, no. 3, pp.755-762.2003.[PubMed]
4.A.-S. Rössler, J.G. Pfaus, H. K. Kia, J. Bernabé,L.Alexandre, and F, Giuliano.*The melanocortin agonist, melanotan ll, enhances proceptive sexual behaviors inthe female rat,”Pharmacol. Biochem.Behav., vol.85, no.3, pp.514-521, NOV.2006.[PubMed]
5.M.R.Safarinejad and S.Y, Hosseini, “Salvage of sildenafil failures withbremelanotide: a randomized, double-blind, placebo controlled study,” J. Urol., vol.179,no.3,pp.1066-1071,Mar.2008.[PubMed]
6.A. H. Clayton et al., “Bremelanotide for female sexual dysfunctions inpremenopausal women: a randomized, placebo-controlled dose-finding trial,Womens Health Lond.Engl., vol.12, no.3, pp.325-337,2016.[PubMed]
7.M.K. Miller, J.R. Smith, J.J. Norman, and A. H. Clayton,“Expert opinion onexisting and developing drugs to treat female sexual dysfunction,” Expert Opin.Emerg.Drugs,vol.23,no.3, pp.223-230,2018.[PubMed]
8.“AMAG Pharmaceuticals and Palatin Technologies Enter Into Exclusive LicensingAgreement for North American Rights to RekyndaTM (bremelanotide), a PotentialTreatment for a Common Female Sexual Disorder -AMAG Pharmaceuticals.[MarketWatchl]
9.H. Ji et al., “The Synthetic Melanocortin (CKPV)2 Exerts Anti-Fungal and Anti.Inflammatory Effects against Candida albicans Vaginitis via Inducing MacrophageM2 Polarization,”PLoS ONE, vol.8, no.2, Feb.2013.[PLOS ONE]
10.V. Maresca, E. Flori, and M. Picardo, “Skin phototype: a new perspective,” PigmentCell Melanoma Res.,vol.28,no.4.pp.378-389,Ju.2015.[PubMedl
11.L. Feller, R. a. G. Khammissa, B. Kramer, M. Altini, and J. Lemmer, “Basal cellcarcinoma, squamous cell carcinoma and melanoma of the head and face.” HeadFace Med., vol.12,p.11,Feb.2016.[PubMed]
11.Clayton AH, Althof SE, Kingsberg S, et al. Bremelanotide for female sexualdysfunctions in premenopausal women: a randomized, placebo-controlled dosefinding trial.Womens Health (Lond).2016;12(3):325-337.doi:10.2217/whe-2016-0018
12.T.R. McMillan, M.A. M. Forster, L.l. Short, A. P. Rudecki, D.L. Cline, and S. L.Gray, “Melanotan ll, a melanocortin agonist, partially rescues the impairedthermogenic capacity of pituitary adenylate cyclase-activating polypeptide deficientmice,Exp.Physiol.,vol.106,no.2,pp.427-437,Feb.2021, doi:10.1113/EP088838.”
13lC. Spana, R. Jordan, and S. Fischkoff, “Effect of bremelanotide on body weight ofobese women: Data from two phase 1 randomized controlled trials,Diabetes ObesMetab., vol.24,no.6,pp.1084-1093, Jun.2022, doi: 10.1111/dom.14672.”
ALL ARTICLES AND PRODUCT INFORMATION PROVIDED ON THIS WEBSITE AREFOR INFORMATONAL AND EDUCATIONAL PURPOSES ONLY.
The products offered on this website are furnished for in-vitro studies only. in-vitro studies(Latin: in glass) are performed outside of the body. These products are not medicines ordrugs and have not been approved by the FDA to prevent, treat or cure any medicalcondition, ailment or disease. Bodily introduction of any kind into humans or animals isstrictly forbidden by law.
Interestingly, PT-141 was pulled from clinical trials before it reached approval for use inwomen suffering from HSDD. This is despite signs that the drug increased the number ofsatisfying sexual events per month and decreased female sexual distress scores in astatistically significant manner without any substantial side effects[5]. Many experts whotreat female sexual dysfunction (FSD) were dismayed to find the peptide was not beingadvanced despite positive results. They point to a lack of established endpoints for trialsof FSD and socio-cultural biases against women’s sexual health as the primaryroadblocks that are inhibiting approval of what they see as much-needed therapies?].They hope that greater attention will be given to the topic and that the FDA will establishmore concrete guidelines for evaluating therapies like PT-141 that can offer benefit.These experts also expressed dismay that the pharmacological treatments were nottested in conjunction with other established means of treating sexual dysfunction as theybelieve that the combination may prove synergistic and that peptides like PT-141 may beuseful for overcoming initial barriers and jump-starting psychological treatment modalities
In 2017, partly in response to the outcry against the cessation of earlier trials, Phase llReconnect trials were launched using subcutaneous injections of PT-141 for FSD. Thenewest version of PT-141, called Rekynda, may soon be available for use in the UnitedStates. lt would be legal to use PT-141 off-label, at that point, to treat both male andfemale sexual dysfunction8. These new trials have relied on the kind of modifiedendpoints that experts in FSD have touted as beneficial to seeing these kinds oftreatments approved.
PT-141 and Hemorrhage
In 2009, PT-141 was modified slightly and investigated as a potential treatment forhemorrhagic shock. Because PT-141 binds to both MC-1R and MC-4R, it reducesischemia and protects tissues against inadequate blood supply in the setting ofhypovolemic (hemorrhagic) shock. The drug, when administered intravenously, does notproduce substantial side effects. lt was last in phase llb trials. The modified version of PT-141 is referred to as PL-6983.
PT-141 and Infection
The MC-1R has been found, in a rat model of a specific fungal infection, to possessimportant anti-fungal and anti-inflammatory properties[9]. This is of particular importancebecause current anti-fungals are limited in terms of their mechanism of action and allproduce serious and treatment-limiting side effects in certain patients. Having analternative to use in the treatment of fungal infections could reduce morbidity andmortality substantially, especially in patients with immune compromise.
PT-141 and Cancer
The MC-1R receptor is an important stimulus of DNA repair pathways and thus is ofinterest in cancer treatment and prevention[1o]. Research shows that people with variantsof MC-1R are at increased risk for both basal cell and squamous cell carcinoma[10].Altered PT-141 may be able to correct the problems experienced as a result of thesevariants and prevent or treat these cancers.Direcciones de investigaciónEn este momento, PT-141 ha recibido una atención intensa y generalizada como tratamiento para la disfunción sexual. Sin embargo, existe una gran cantidad de investigaciones potenciales fuera de la disfunción sexual y la hemorragia a las que se podría aplicar el PT-141. Por ejemplo, se sabe que el MC-4R está defectuoso o falta en ciertos casos de obesidad y puede representar hasta el 6% de todos los casos de obesidad de aparición temprana. PT-141 ofrece un medio único para explorar esta causa particular de la obesidad y potencialmente ilustrar una vía de intervención. MC-1R desempeña un papel tanto en el dolor como en la inflamación, así como en la patología renal y la propagación de infecciones. Hay una gran cantidad de investigaciones disponibles sobre las que PT.141 podría ayudar a arrojar luz.
PT-141 presenta efectos secundarios mínimos, baja biodisponibilidad oral y subcutánea excelente en ratones. La dosis por kg en ratones no se ajusta a la de los humanos. El PT-141 a la venta en PeptideSciences se limita únicamente a la investigación científica y educativa, no al consumo humano. Compre PT-141 únicamente si es un investigador autorizado.Autor del artículoLa literatura anterior fue investigada, editada y organizada por el Dr. Logan, M.D. El Dr. Logar tiene un doctorado de la Facultad de Medicina de la Universidad Case Western Reserve y una licenciatura. en biología molecular.Autor de revista científicaDr. Sheryl A. Kingsberg is the chief of behavioral medicine at University Hospitals CaseMedical Center and professor in Reproductive Biology and Psychiatry at Case WesternReserve University. Her areas of clinical specialization include sexual medicine, femalesexual disorders, cognitive behavioral psychotherapy, menopause, pregnancy andpostpartum mood disorders, psychological aspects of infertility, and psychological andsexual aspects of cancer. Dr. Kingsberg’s primary research interests are in treatments forfemale sexual disorders and the psychological aspects of infertility and menopause. Sheled a randomized, placebo-controlled dose-finding trial for PT-141. She has numerouspublications in many national and international journals, sits on the editorial board ofMenopause and has authored numerous chapters on topics including perimenopauseand sexuality, oocvte donation, infertility and aging, the treatment of psychogenic erectiledysfunction and sexuality after cancer. Dr. Kingsberg received her PhD from theUniversity of South Florida in Tampa and completed her fellowshin in sexual medicine aUniversity Hospitals Case Medical Center, She is an active member in a number ofnational and international organizations including the American Psychological Associationand the American Society for Reproductive Medicine. She currently sits on the Board ofTrustees of The North American Menopause Society, and serves as the current treasurerof the Society for Assisted Reproductive Technologies. Dr. Kingsberg s a past president otThe International Society for the Study of Women’s Sexual Health.
Dr. Sheryl A. Kingsberg is being referenced as one of the leading scientists involved inthe research and development of PT-141, in no way is this doctor/scientist endorsing oradvocating the purchase, sale, or use of this product for any reason, There is no affiliationor relationship, implied or otherwise, between PEPTIDE GURUS and this doctor. Thepurpose of citing the doctor is to acknowledge, recoanize, and credit the exhaustiveresearch and development efforts conducted by the scientists studying this peptide. DrKingsbera is listed in [12l under the referenced citations.Citas referenciadas1.M.Sandrock,A.Schulz,C.Merkwitz,T.Schüneberg, K.Spanel-Borowski, and A.Ricken, “Reduction in corpora lutea number in obese melanocortin-4-receptor-deficient mice,”Reprod. Biol. Endocrinol.RBE,vol.7. p.24, Mar. 2009.
2.R.C.Rosen,L.E.Diamond, D.C.Earle,A. M. Shadiack, and P. B. MolinoffEvaluation of the safety, pharmacokinetics and pharmacodynamic effects ofsubcutaneously administered PT-141, a melanocortin receptor agonist, in healthymale subjects and in patients with an inadeguate response to Viagra,” int. J. lmpot.Res., vol.16,no.2,pp.135-142,Apr.2004.[PubMed]
3.H.Wessells,V.. Hruby, J.Hackett, G. Han, P. Balse-Srinivasan, and T. W.Vanderah,“Ac-Nle-c[Asp-His-DPhe-Arg-Trp-Lys]-NH2 induces penile erection viabrain and spinal melanocortin receptors,”Neuroscience, vol. 118, no. 3, pp.755-762.2003.[PubMed]
4.A.-S. Rössler, J.G. Pfaus, H. K. Kia, J. Bernabé,L.Alexandre, and F, Giuliano.*The melanocortin agonist, melanotan ll, enhances proceptive sexual behaviors inthe female rat,”Pharmacol. Biochem.Behav., vol.85, no.3, pp.514-521, NOV.2006.[PubMed]
5.M.R.Safarinejad and S.Y, Hosseini, “Salvage of sildenafil failures withbremelanotide: a randomized, double-blind, placebo controlled study,” J. Urol., vol.179,no.3,pp.1066-1071,Mar.2008.[PubMed]
6.A. H. Clayton et al., “Bremelanotide for female sexual dysfunctions inpremenopausal women: a randomized, placebo-controlled dose-finding trial,Womens Health Lond.Engl., vol.12, no.3, pp.325-337,2016.[PubMed]
7.M.K. Miller, J.R. Smith, J.J. Norman, and A. H. Clayton,“Expert opinion onexisting and developing drugs to treat female sexual dysfunction,” Expert Opin.Emerg.Drugs,vol.23,no.3, pp.223-230,2018.[PubMed]
8.“AMAG Pharmaceuticals and Palatin Technologies Enter Into Exclusive LicensingAgreement for North American Rights to RekyndaTM (bremelanotide), a PotentialTreatment for a Common Female Sexual Disorder -AMAG Pharmaceuticals.[MarketWatchl]
9.H. Ji et al., “The Synthetic Melanocortin (CKPV)2 Exerts Anti-Fungal and Anti.Inflammatory Effects against Candida albicans Vaginitis via Inducing MacrophageM2 Polarization,”PLoS ONE, vol.8, no.2, Feb.2013.[PLOS ONE]
10.V. Maresca, E. Flori, and M. Picardo, “Skin phototype: a new perspective,” PigmentCell Melanoma Res.,vol.28,no.4.pp.378-389,Ju.2015.[PubMedl
11.L. Feller, R. a. G. Khammissa, B. Kramer, M. Altini, and J. Lemmer, “Basal cellcarcinoma, squamous cell carcinoma and melanoma of the head and face.” HeadFace Med., vol.12,p.11,Feb.2016.[PubMed]
11.Clayton AH, Althof SE, Kingsberg S, et al. Bremelanotide for female sexualdysfunctions in premenopausal women: a randomized, placebo-controlled dosefinding trial.Womens Health (Lond).2016;12(3):325-337.doi:10.2217/whe-2016-0018
12.T.R. McMillan, M.A. M. Forster, L.l. Short, A. P. Rudecki, D.L. Cline, and S. L.Gray, “Melanotan ll, a melanocortin agonist, partially rescues the impairedthermogenic capacity of pituitary adenylate cyclase-activating polypeptide deficientmice,Exp.Physiol.,vol.106,no.2,pp.427-437,Feb.2021, doi:10.1113/EP088838.”
13lC. Spana, R. Jordan, and S. Fischkoff, “Effect of bremelanotide on body weight ofobese women: Data from two phase 1 randomized controlled trials,Diabetes ObesMetab., vol.24,no.6,pp.1084-1093, Jun.2022, doi: 10.1111/dom.14672.”
ALL ARTICLES AND PRODUCT INFORMATION PROVIDED ON THIS WEBSITE AREFOR INFORMATONAL AND EDUCATIONAL PURPOSES ONLY.
The products offered on this website are furnished for in-vitro studies only. in-vitro studies(Latin: in glass) are performed outside of the body. These products are not medicines ordrugs and have not been approved by the FDA to prevent, treat or cure any medicalcondition, ailment or disease. Bodily introduction of any kind into humans or animals isstrictly forbidden by law.
PeptideGurus es un proveedor líder de péptidos de investigación fabricados en Estados Unidos y ofrece productos de alta calidad a precios competitivos. Con un enfoque en la excelencia y el servicio al cliente, garantizan un proceso de pedido seguro y conveniente con envío global.
CONTACTO